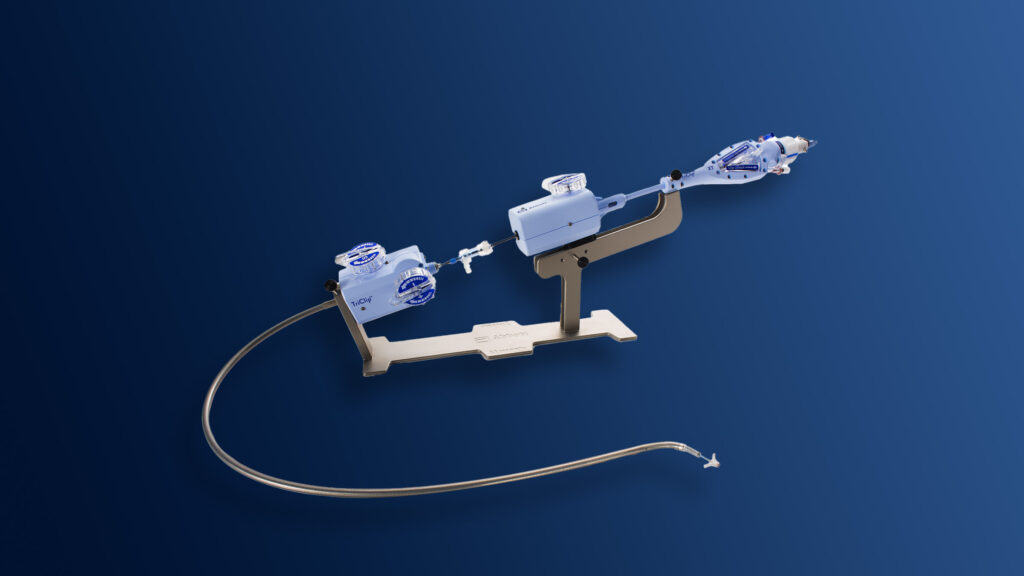
An advisory panel to the Food and Drug Administration voted in favor of a new device from Abbott meant to treat patients with tricuspid heart valve disease. All but one of the 14 panelists said the treatment’s benefits outweighed its risks. The FDA tends to follow advisory panel recommendations.
“If we can help symptoms with this at a low cost in terms of risk, I think it makes a lot of sense,” said James Blankenship, a panelist and cardiologist at the University of New Mexico.
advertisement
The device is called the TriClip, and it addresses a disease called tricuspid regurgitation: a heart condition that causes blood to leak backward through the tricuspid heart valve. The condition impacts 1.6 million people in the United States. Symptoms include fatigue, swelling, and atypical heart rhythms. In severe cases, the condition can lead to heart failure.
Get unlimited access to award-winning journalism and exclusive events.